Hi! This is my news feed. I will be adding posts to this feed for your interest. Kind regards, Paul
GlucoRegulate vs Tirzepatide in obesity
Dietary supplements are often taken to support good health. It’s rare to see evidence of comparison with leading pharmaceuticals in clinical trial. An example is weight loss drug Tirzepatide (Zepbound) and supplement GlucoRegulate. Tirzepatide (5 mg, once weekly subcutaneous injection) for 72 weeks to subjects living with obesity improved insulin sensitivity, measured by the oral glucose insulin sensitivity (OGIS) index, by 75 units – including diet counselling to achieve 500 calories per day decrease and at least 150 min per week exercise; SURMOUNT-1 trial. Dietary supplement GlucoRegulate (once daily, oral capsule) for 8 weeks to subjects living with obesity improved OGIS index by 58 units – with no change in diet and exercise; HATFF trial. A similar improvement in metabolic health with GlucoRegulate as for Tirzepatide and achieved over a shorter period with minimal lifestyle change and low cost to consumer and healthcare provider. A remarkable comparison indeed! Increased insulin sensitivity in obesity is important to counter insulin resistance for improved metabolic, heart, liver, kidney and respiratory health. GlucoRegulate is a supplement by GloVitality (UK) Ltd (https://glovitality.com/glucoregulate/).

Supporting papers: Mounjaro – Mari et al., Diabetes Care 2025; 48: 1622–1627; GlucoRegulate (then called Glo1 inducer) – Xue at al., Diabetes 2016; 65: 2282–2294.
Paul J Thornalley 19th March 2026
Insulin resistance
Insulin resistance is the impaired response to insulin of tissues that are normally insulin-responsive. It is increasingly recognized as impacting the liver, skeletal muscle, adipose tissue and also the brain. The mechanisms are distinct in different tissues. It is prevalent in obesity, prediabetes and type 2 diabetes and is often the primary initiator leading to type 2 diabetes development.
Hepatic insulin resistance is so-called “mixed” insulin resistance and is due to down regulation of insulin receptor substrate 2 (IRS-2). A major consequence of this is impaired down regulation of gluconeogenesis in the post-meal absorptive phase leading to hypersecretion of glucose from the liver. It is caused by overactivation of carbohydrate-response element binding protein (ChREBP) by a sugar rich diet, hyperinsulinaemia and also multilayered epigenetic and extrahepatic mechanisms, including regulation by the central nervous system and glucagon. Involvement of ChREBP is driven by glucokinase-linked glycolytic overload in hepatocytes. It is likely reversed by GlucoRegulate supplement, trans-resveratrol with hesperetin (Rabbani and Thornalley, Clinical Science 139, 1 – 25, 2025).
Peripheral insulin resistance is the impaired response of skeletal muscle and adipose tissue to insulin. Normally in the absorptive phase insulin stimulates uptake of glucose into muscle for glycogen synthesis and uptake of glucose into adipose tissue for synthesis of lipids. Impaired recruitment of insulin-dependent GLUT4 glucose transporter appears to have a key role. We have described how hexokinase-2 linked glycolytic overload in the fasting period during impaired fasting glucose with increased fasting glucose concentration contributes to this. It is corrected by GlucoRegulate supplement (Rabbani and Thornalley, Front. Endocrinol. 14: 1268308, 2024).
Brain insulin resistance may be inferred from decreased response amplitudes to auditory and visual stimuli using electroencephalography and magneto-encephalography during a hyperinsulinemic euglycemic clamp. It is typically found in subjects living with obesity and type 2 diabetes which also have hepatic and peripheral insulin resistance. It is associated with decreased memory and improved cognitive performance and is thought to be mediated by inflammation and oxidative stress (Milstein, J.L., and Ferris, H.A. Mol Metab 52, 101234, 2021). It is likely corrected by GlucoRegulate supplement which is currently under consideration.
Paul J Thornalley, 15th March 2026
Happy New Year 2026

Happy New Year! Looking forward to working with in-house and collaborating international teams in 2026 to advance projects in biomedical research and commercialization – particularly of precision dietary/food supplement GlucoRegulate (through Glovitality Ltd and Glocentrica Ltd) and screening blood test for autism (through iDiagnostix Ltd).
“Come, my friends, ‘Tis not too late to seek a newer world”. From Ulysses, Alfred Lord Tennyson.
Paul J Thornalley, 1st January 2026
What have we discovered in 2025?
- Diets rich in simple sugars and/or fats provide a gateway to metabolic dysfunction associated steatotic liver disease (MASLD) and supplement GlucoRegulate is deserving of evaluation of its early-stage treatment. Link
- Diets rich in glucose and fructose produce glycolytic overload in the liver, inducing mixed insulin resistance and providing the gateway for the path to development of type 2 diabetes. Link
- The primary metabolic effect of fructose in the liver is to lift the restriction on flux of glucose metabolism by glucokinase regulatory protein (GKRP) and thereby facilitate glycolytic overload in the liver. Link
- Hexokinase-2 linked glycolytic overload mediates diabetic embryopathy in an experimental model where supplement GlucoRegulate provides preventive therapy. Link
- GlucoRegulate is a multi-modal dietary supplement primarlity acting through activation of Nrf2 with downstream health benefits linked to prevention of dicarbonyl stress, lipid peroxidation, oxidative stress, proteotoxicity and hyperglycemia-linked glycolytic overload. Downstream benefits were improved regulation of glucose and lipid metabolism and decreased inflammation, extracellular matrix remodeling and senescence markers. Link
Still much more to discover in 2026!
Paul J Thornalley, 26th December 2025.
New paper
A pleasure to share this paper covering dietary glucose- and fructose-driven mechanisms of development of MASLD and in so doing, revealing the mechanism of development of insulin resistance and processes leading to peripheral insulin resistance and type 2 diabetes.
Summary points
- Experimental and clinical evidence suggest pathogenic mechanisms linked to abnormal metabolism of glucose and fructose in the liver contribute to the initiation and development of metabolic dysfunction-associated steatotic liver disease (MASLD).
- Our aim was to review these and consider if the hypothesis of glycolytic overload applies. We found that it does with regulation of glucose metabolism in hepatocytes overwhelmed by sugar-rich diets and hyperglycemia in prediabetes and diabetes, where flux of glucose metabolism in the absorptive phase is predicted to increase up to 4-fold.
- Related increased glucose-6-phosphate, fructose-6-phosphate and methylglyoxal activate transcription factors, metabolic pathways and stress response signalling producing hepatic insulin resistance, steatosis, inflammation and fibrosis.
- Key mediators are: carbohydrate response element binding protein (ChREBP), hexosamine pathway, and methylglyoxal-activated unfolded protein response.
- Maintained insulin receptor substrate-1 (IRS-1) signaling increases the sterol response element binding protein-1c lipogenic response; whereas down-regulated IRS-2 signaling impairs suppression of hepatic glucose production.
- Hepatocytes then export glucose excessively, increasing fasting plasma glucose and risk of peripheral insulin resistance, type 2 diabetes and vascular complications.
- Activators of nuclear factor erythroid 2-related factor 2 (Nrf2) provide a novel strategy for therapy, diverting excess glucose metabolism to the pentosephosphate pathway, decreasing methylglyoxal and suppressing lipogenesis.
- Nrf2 activator, trans-resveratrol and hesperetin combination (GlucoRegulate), corrected glycolytic overload and insulin resistance clinically and now merits evaluation for treatment of early-stage MASLD.
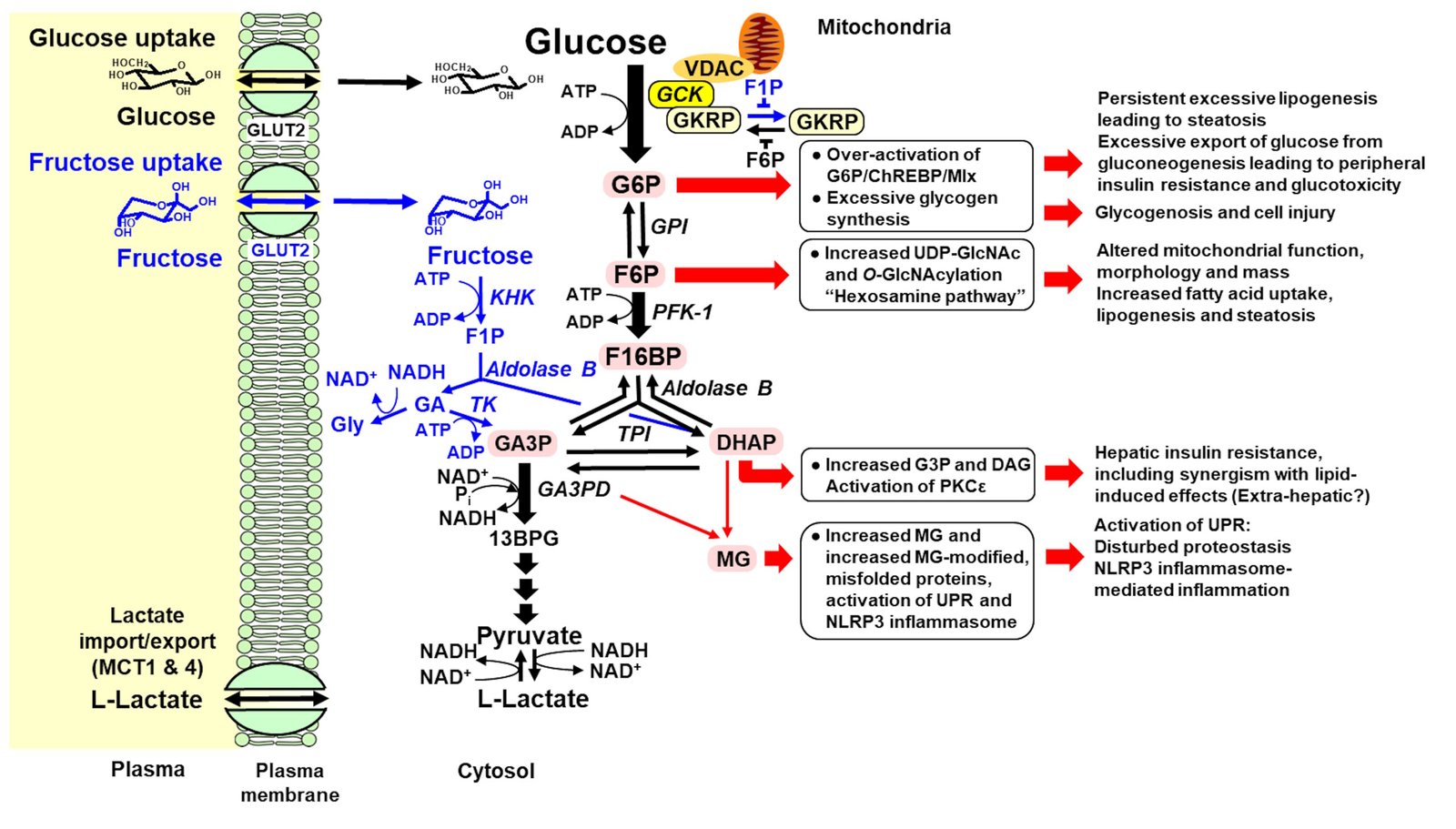
Figure 1: Unscheduled glycolysis and glycolytic overload in the initiation of metabolic dysfunction-associated steatotic liver disease. Key: pink back-filled metabolites, glycolytic intermediates increased in unscheduled glycolysis; red arrows – pathways of pathogenesis initiated by unscheduled glycolysis in hepatocytes; and blue metabolites and arrows – metabolism of fructose. Names of enzymes are given in italics. The nuclear translocation of GCK and GKRP is not shown for clarity.
Paul J Thornalley, 7th November 2025
Clinical evidence-based dietary supplements – appropriately considered along with drugs for adjunct and first line treatments
I saw a paper today on a study finding that maintaining endothelial cell levels of NADPH is a strategy to counter vascular aging. Having found this, drug repurposing was explored for 1419 drugs. However, we already know that GlucoRegulate supplement increases expression of glucose-6-phosphate dehydrogenase (G6PD) to increase NADPH and is well tolerated and safe. See our study of GlucoRegulate development. This is an example where dietary supplements targeting activation of Nrf2 and increased expression of antioxidant response element-linked G6PD can be considered along with repurposed drugs as adjunct or even first line treatments – particularly as dietary supplements typically do not suffer the clinical adverse effects of synthetic drugs. The dietary supplements need to be clinically evidence-based, of course, and if used in special drug delivery format or out of the known safe and well-tolerated range, assessed for safety.
New paper
Plasma Glycated and Oxidized Amino Acid-Based Screening Test for Clinical Early-Stage Osteoarthritis
Aisha Nasser J. M. Al-Saei, Usman Ahmed, Edward J. Dickenson, Kashif Rajpoot, Mingzhan Xue, Essam M. Abdelalim, Abdelilah Arredouani, Omar M. E. Albagha, Damian R. Griffin, Paul J. Thornalley and Naila Rabbani
The diagnosis of early-stage osteoarthritis (eOA) is important in disease management and outcomes. Herein we report the clinical validation of a blood test for the diagnosis of eOA in a large patient cohort using trace-level glycated and oxidized amino acid analytes. Subjects were recruited and enrolled in two study groups: subjects with eOA of the hip (n = 110) and asymptomatic controls (n = 120). Their plasma was analyzed for glycated and oxidized amino acids by quantitative liquid chromatography–tandem mass spectrometry. Algorithms were developed using plasma hydroxyproline and 12 glycated and oxidized amino acid analyte features to classify the subjects with eOA and asymptomatic controls. The accuracy was defined as the percentage of the subjects correctly classified in the test set validation. The minimum number of analyte features required for the optimum accuracy was five glycated amino acid analytes: Nω-carboxymethyl-arginine, hydroimidazolones derived from glyoxal, methylglyoxal and 3-deoxyglucosone, and glucosepane. The classification performance metrics included an accuracy of 95%, sensitivity of 96%, specificity of 94%, area under the curve of the receiver operating characteristic curve of 99%, and positive and negative predictive values of 94% and 97%. We concluded that an assay of five trace-level glycated amino acids present in plasma can provide a simple blood test for the screening of eOA. This is predicted to improve the case identification for expert referral 9-fold.
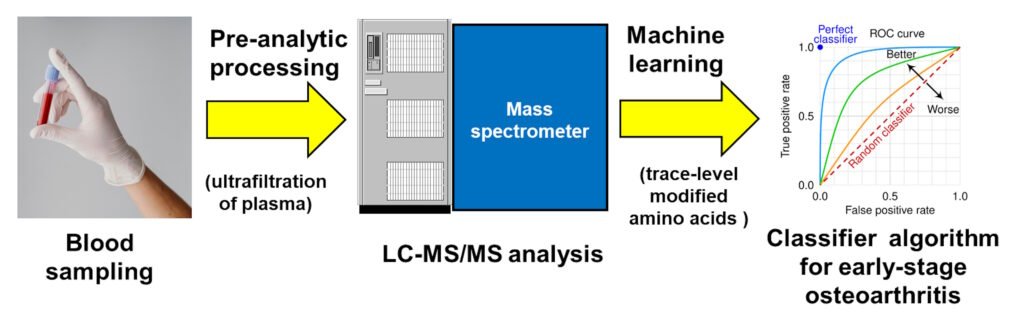
Antioxidants 2025, 14, 1146. https://doi.org/10.3390/antiox14101146
Published 23rd September 2025.
New Paper
Glyoxalase 1 Inducer, trans-Resveratrol and Hesperetin–Dietary Supplement with Multi-Modal Health Benefits
by Mingzhan Xue, Naila Rabbani and Paul J. Thornalley
Abstract
A dietary supplement, trans-resveratrol and hesperetin (tRES+HESP)—also known as GlucoRegulate—induces increased expression of glyoxalase 1 (Glo1) by activation of transcription factor Nrf2, countering accumulation of the reactive dicarbonyl glycating agent, methylglyoxal. tRES+HESP corrected insulin resistance and decreased fasting and postprandial plasma glucose and low-grade inflammation in overweight and obese subjects in a clinical trial. The aim of this study was to explore, for the first time, health-beneficial gene expression other than Glo1 induced by tRES+HESP in human endothelial cells and fibroblasts in primary culture and HepG2 hepatoma cell line and activity of cis-resveratrol (cRES) as a Glo1 inducer. We measured antioxidant response element-linked gene expression in these cells in response to 5 µM tRES+HESP by the NanoString method. tRES+HESP increases gene expression linked to the prevention of dicarbonyl stress, lipid peroxidation, oxidative stress, proteotoxicity and hyperglycemia-linked glycolytic overload. Downstream benefits were improved regulation of glucose and lipid metabolism and decreased inflammation, extracellular matrix remodeling and senescence markers. The median effective concentration of tRES was ninefold lower than cRES in the Glo1 inducer luciferase reporter assay. The GlucoRegulate supplement provides a new treatment option for the prevention of type 2 diabetes and metabolic dysfunction–associated steatotic liver disease and supports healthy aging.
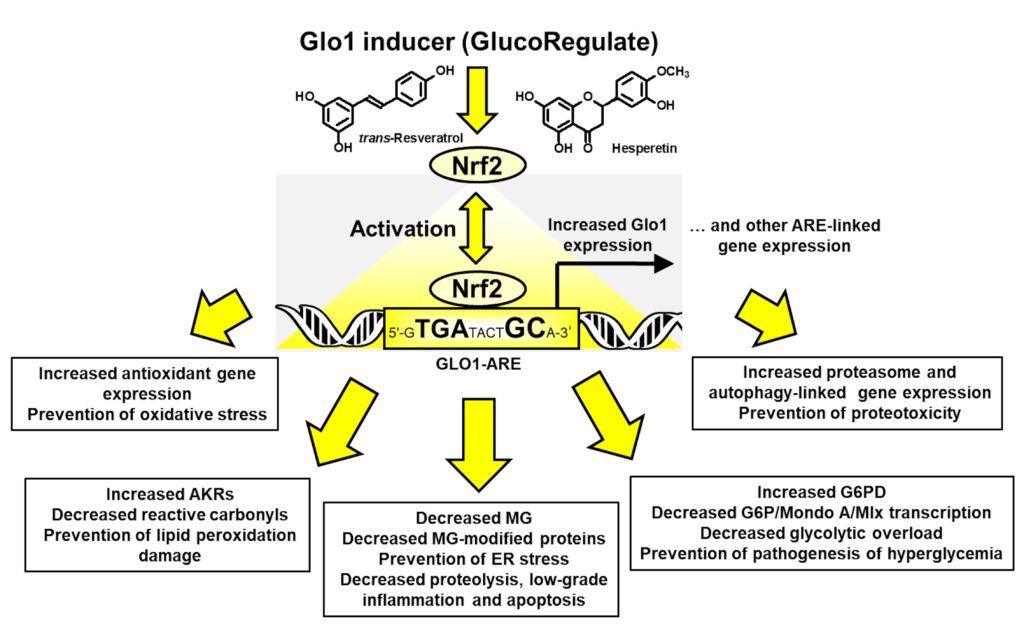
Keywords: methylglyoxal; oxidative stress; lipid peroxidation; ER stress; proteotoxicity; insulin resistance; diabetes; MASLD; aging; resveratrol
Antioxidants 2025, 14(8), 956; https://doi.org/10.3390/antiox14080956
Published: 4 August 2025
New paper
Overload of Glucose Metabolism as Initiating Factor in Diabetic Embryopathy and Prevention by Glyoxalase 1 Inducer Dietary Supplement
by Parri Wentzel, Mingzhan Xue, Naila Rabbani, Ulf J. Eriksson, and Paul J. Thornalley
Abstract
Hyperglycemia in early-stage embryogenesis is linked to diabetic embryopathy. High-glucose-concentration-induced accumulation of hexokinase-2 (HK2) may initiate metabolic dysfunction that contributes to diabetic embryopathy, including increased formation of methylglyoxal (MG). In this study, we evaluated changes in HK2 protein levels and embryo dysmorphogenesis in an experimental model of diabetic embryopathy. Rat embryos were cultured with high glucose concentrations, and the effects of glyoxalase 1 (Glo1) inducer, trans-resveratrol and hesperetin (tRES + HESP) were evaluated. Rat embryos, on gestational day 9, were cultured for 48 h in low and high glucose concentrations with or without tRES + HESP. Embryo crown–rump length, somite number, malformation score, concentrations of HK2 and Glo1 protein, rates of glucose consumption, and MG formation were assessed. Under low-glucose conditions, embryos exhibited normal morphogenesis. In contrast, high-glucose conditions led to reduced crown–rump length and somite number, and an increased malformation score. The addition of 10 μM tRES + HESP reversed these high glucose-induced changes by 60%, 49%, and 47%, respectively. Embryos cultured in high glucose showed increases in HK2 concentration (42%), glucose consumption (75%), and MG formation (27%), normalized to embryo volume. These elevated HK2 levels were normalized by treatment with 10 μM tRES + HESP. Thus, high-glucose-induced metabolic dysfunction and embryopathy may both be initiated by HK2 accumulation and may be preventable with tRES + HESP treatment.
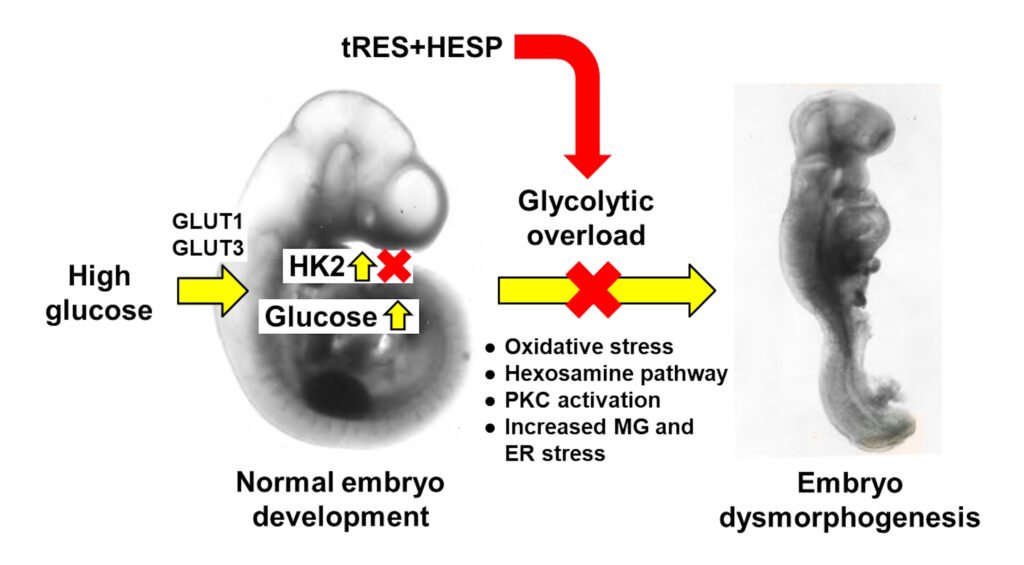
Keywords: hyperglycemia; metabolic dysfunction; glycation; diabetic embryopathy; teratogenesis
Antioxidants 2025, 14(8), 1022; https://doi.org/10.3390/antiox14081022
Published: 21st August 2025
Conference presentation
XIII annual conference – Autism: Challenges and Solutions (ANO), Moscow, Russia, 12th – 14th September 2025

Title of presentation: Maternal and infant hyperglycemia – emerging evidence of impact on risk and symptoms of autism
Naila Rabbani and Paul J Thornalley
Summary
- Maternal obesity, gestational diabetes and diabetes type 1 and type 2 increases risk of autism in offspring by 42%, 48% and 74%, respectively
- This is likely due to exposure of the developing embryo and fetus to high glucose concentration in insulin resistance and hyperglycemia
- Abnormal high glucose metabolism occurs in regions of the brain with hexokinase 2 – susceptible to hexokinase-2 linked glycolytic overload; cf. in diabetic embryopathy
- This is expected to cause dysfunction in oligodendrocyte precursors, astrocytes and microglia affecting neuronal activity and immune responses
- Insulin resistance in infants with autism may sustain and prolong symptoms
- Improved glycemic health of mother and infant may reduce risk of autism in offspring and severe symptoms in children. Supplement treatment may soon emerge
Paul J Thornalley, 16th September 2025